ClariVein®OC is a specialty infusion catheter used for occlusion of incompetent veins in patients with superficial venous reflux.
clariVein®OC 是一種特殊的輸液導管,用於閉塞淺靜脈逆流患者的功能不全的靜脈。
ClariVein®OC 는 제 기능을 발휘하지 못하는 표재성 정맥 역류 환자의 정맥 폐색에 사용하는 특수 주입 카테터입니다.
ClariVein®OC is designed for easy insertion and patient comfort and enables patients to quickly return to their work and normal activities.
clariVein®OC 設計低調,方便進入,讓患者感到舒適,從而促進快速恢復工作和正常活動。
ClariVein®OC 는 진입이 용이하고 환자가 편안하도록 설계되어 업무와 일상 활동으로 빨리 복귀할 수 있도록 촉진합니다.
What is Varicose Veins?
什麼是靜脈曲張?
하지정맥류란 무엇인가요?
Varicose veins are enlarged, bulging veins appearing on the skin surface of the legs.
The underlying cause of varicose veins is superficial venous reflux, which occurs when the tiny valves inside the leg veins that control blood flow from the legs to the heart become damaged.
Such damaged valves can cause various symptoms including pain and can sometimes cause unsightly varicose veins. If left untreated, they can progress to superficial venous reflux disease to cause other more serious circulatory and skin problems extending beyond the legs.
靜脈曲張是腿部皮膚表面出現的擴大、凸出的靜脈。 靜脈曲張的根本原因是靜脈逆流病,當腿部靜脈內控制血液從腿部流向心臟的微小瓣膜受損時,就會發生靜脈曲張
如果不及時治療,除了導致難看且有時疼痛的靜脈曲張外,靜脈逆流疾病還可能發展為腿部其他更嚴重的循環和皮膚問題。
하지정맥류는 다리의 피부 표면에 정맥이 늘어나고 돌출되어 보이는 것입니다.
하지정맥류의 근본 원인은 표재성 정맥 역류 질환으로, 이는 다리에서 심장으로 향하는 혈류를 조절하는 다리 정맥 내부의 작은 판막이 손상될 때 발생합니다.
이러한 판막의 손상은 고통을 포함한 여러 증상을 유발하고 때로는 미관상 좋지 않은 하지정맥류를 유발하게 되고, 치료하지 않고 방치하면 표재성 정맥 역류 질환이 진행되어 다리에 이외의 더 중요한 순환계 및 피부까지 문제를 일으킬 수 있습니다.
What happens if varicose veins are left untreated?
如果我不治療靜脈曲張會發生什麼事?
하지정맥류를 치료하지 않으면 어떻게 되나요?
Varicose veins do not improve without treatment. Varicose veins can worsen if they are left untreated. Today, there are various treatments available for treating varicose veins with virtually no pain and patients can quickly return to normal life.
不幸的是,如果不介入,這種疾病就不會好轉。 如果靜脈曲張不及時治療,這種疾病可能會使人衰弱,甚至可能惡化。 然而,好消息是,現在有一些治療方法可以有效解決靜脈曲張,而且疼痛很少,讓您很快恢復正常生活。
하지정맥류는 치료 없이는 나아지지 않습니다. 하지정맥류를 치료하지 않고 방치하면 더 악화될 수 있습니다. 오늘날에는 거의 통증 없이 하지정맥류를 치료할 수 있는 다양한 치료법이 있어 일상으로 빠르게 돌아갈 수 있습니다.
What kind of treatments are available?
有哪些治療選擇?
어떠한 치료법이 있나요?
All procedures have the same goal. That is to remove or close the varicose veins. The purpose is to create normal veins in the legs that will naturally return blood from the legs to the heart.
In the past, the most common type of varicose vein treatment was an operation called ‘Stripping,’ which involves complete removal of the incompetent veins with great pain. It had been considered to be the gold standard of care until early 2000s when the minimally invasive thermal treatment was introduced.
Currently, MOCA (Mechanical Occlusion Chemically Assisted) is recommended. ClariVein OC catheter is used to treat the root cause of malfunctioning veins of superficial venous reflux patients. In studies, ClariVein OC showed multiple advantages such as reduced pain, quick return to work and normal daily activities, etc.1
所有手術都有相同的目標:透過切除或閉合曲張靜脈,血液自然會採用替代路徑流向腿部未受影響的靜脈,進而接管血液從腿部返回心臟的任務。
先前,靜脈曲張最常見的治療方法是一種稱為「靜脈剝離」的痛苦手術,該手術在手術室中進行,完全切除有問題的靜脈。 這一直是黃金標準,直到 2000 年代初微創熱技術作為替代護理標準出現為止。
今天,您的醫生提供 MOCA(機械消融,化學輔助)。使用 clariVein®OC 導管用於治療淺層靜脈逆流患者靜脈功能不全的根本原因。研究表明 clariVein®OC 具有許多優點,包括減輕疼痛、更快恢復工作和正常活動。1
모든 시술의 목표는 동일합니다. 하지정맥류를 제거하거나 폐쇄하는 것입니다. 혈액이 자연스럽게 다리에서 심장으로 되돌려 보내는 다리의 정상 정맥을 만드는 것이 목적입니다.
예전의 가장 일반적인 하지정맥류 치료법은 수술실에서 문제가 되는 정맥을 완전히 제거하는 ‘Stripping’ 이라는 많은 통증이 따르는 수술이었습니다. 당시에는 본 수술이 최적의 표준 치료였으나 2000년대 초반에 열을 이용한 최소 침습적 법이 대체 표준 치료로 등장했습니다.
오늘날에는 MOCA(기계화학정맥폐쇄술)를 제안합니다. ClariVein OC 카테터를 사용하여 표재성 정맥 역류 환자의 제 기능을 발휘하지 못하는 정맥의 근본 원인을 치료합니다. ClariVein OC가 통증 감소, 조속한 업무 및 일상 활동 복귀 등 장점이 많은 것으로 연구에서 밝혀져 있습니다.1
What is ClariVein®OC?
什麼是ClariVein®OC?
ClariVein®OC란 무엇인가요?
ClariVein®OC is a specialty infusion catheter with a 360-degree rotating wire tip designed for the 360-degree dispersion of physician-specified agents (sclerosant) to the targeted treatment area.
ClariVein®OC is a small, thin catheter (tube) that physicians use in vascular treatments. ClariVein®OC is remarkably smaller than other devices allowing the vascular entry point to be smaller.
clariVein®OC 是一種附旋轉線尖的專科輸注導管,設計用於將醫生指定的藥劑以受控方式 360 度分散到目標治療區域。
clariVein®OC 裝置是一種細長的導管(管),醫生透過針大小的入口點將其暫時插入外周脈管系統。 clariVein®OC 比周邊血管治療中使用的其他裝置小幾倍,允許更小的入口點。
ClariVein®OC는 360도로 회전하는 도관 끝부분에 의사가 지정한 약제(경화제) 를 치료 표적 부위에 360도 분산시킬 수 있도록 설계된 특수 주입 카테터입니다.
ClariVein®OC는 의사가 혈관에 사용하는 작고 얇은 도관(Tube)입니다. ClariVein®OC는 다른 장치보다 현저히 작아서 혈관에 삽입되는 입구가 더 작습니다.
Why ClariVein®OC?
為什麼選擇 ClariVein®OC?
왜 ClariVein®OC를 선택해야 하나요?
ClariVein®OC is simpler and quicker than other minimally invasive treatments. Studies also showed that treatments with ClariVein®OC is up to 74% less painful than other minimally invasive peripheral vascular treatments.1 It does not use thermal energy and therefore tumescent anesthesia using multiple injections across the treatment area is not required. This shortens the procedure time, reduces pain and discomfort, and causes no bruising.
Please contact your physician to find if ClariVein®OC is right for you.
Consult your physician about various benefits that can be provided by ClariVein®OC compared with other treatments.
使用的程式 clariVein®OC 通常更簡單、更快,研究顯示比其他微創週邊血管治療方法可減輕高達 74% 的疼痛1。 由於不使用熱能,因此無需沿著治療區域的長度多次針刺注射麻醉(止痛)藥物。 這可以縮短手術時間,減少疼痛和不適,並消除瘀傷。
詢問您的醫生是否 clariVein®OC 適合您
向您的醫生詢問兩者之間的區別 clariVein®與其他可用的治療方法相比,OC 閉塞導管可以提供更好的效果。
ClariVein®OC 다른 최소침습치료법 간소하고 빠르며, 연구에 따르면 다른 최소 침습 말초 혈관 치료법보다 통증이 최대 74% 적은 것으로 나타났습니다.1. 열 에너지를 사용하지 않기 때문에 치료 부위 전체에 여러 번의 주사를 이용한 팽창마취가 필요가 없습니다. 이를 통해 시술 시간이 단축되고, 통증과 불편함이 줄어들며, 멍이 생기지 않습니다.
ClariVein®OC가 본인에게 적합한지 담당 의사에게 문의하세요.
다른 치료법과 비교했을 때 ClariVein®OC가 제공할 수 있는 다양한 장점에 대해 담당 의사와 상의하세요.
How does the procedure work?
手術過程中會發生什麼事?
시술이 어떻게 진행되나요?
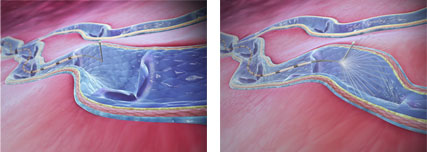
Once the catheter is inserted percutaneously into the affected vein, the rotating tip of ClariVein®OC delivers agents and treats the inside of the veins. Agents are delivered through the rotating tip of the catheter and can be dispersed 360-degree in the vasculature.
This procedure does not take long time and patient discomfort is also minimized.
一旦使用針大小的入口穿過皮膚放置在外周脈管系統內,旋轉尖端 clariVein®OC 啟動以治療輸送醫生指定藥物的血管內部。 藥物透過導管獨特的旋轉尖端輸送,可 360 度覆蓋血管。
該過程通常需要很短的時間,且造成的不適也很小。
문제가 있는 정맥에 피부를 통해 카테터를 삽입 후 , ClariVein®OC의 회전하는 끝부분이 회전하면서 약물을 전달하여 혈관 내부를 치료합니다. 카테터의 회전하는 끝부분을 통해 약물이 전달되어 혈관에 약물을 360도로 분산할 수 있습니다.
이 절차는 시간이 많이 걸리지 않으며 환자의 불편함도 최소화합니다.
What happens after the procedure?
Please follow your doctor’s instructions for care after your procedure. Patients can expect a quick return to normal daily activities.
手術後請遵循醫生的照護指示。 在某些情況下,預計可迅速恢復正常活動。
시술 후 관리에 대해서는 담당 의사의 지시를 따르십시오. 일상 활동으로의 빠른 복귀를 기대할 수 있습니다.
1. R. van Eekeren, et al., Postoperative Pain and Early Quality of Life After Radiofrequency Ablation and Mechanochemical Endovenous Ablation of Incompetent Great Saphenous Veins, journal of vascular surgery, Volume 57, Number 2, February 2013, p. 445-450.